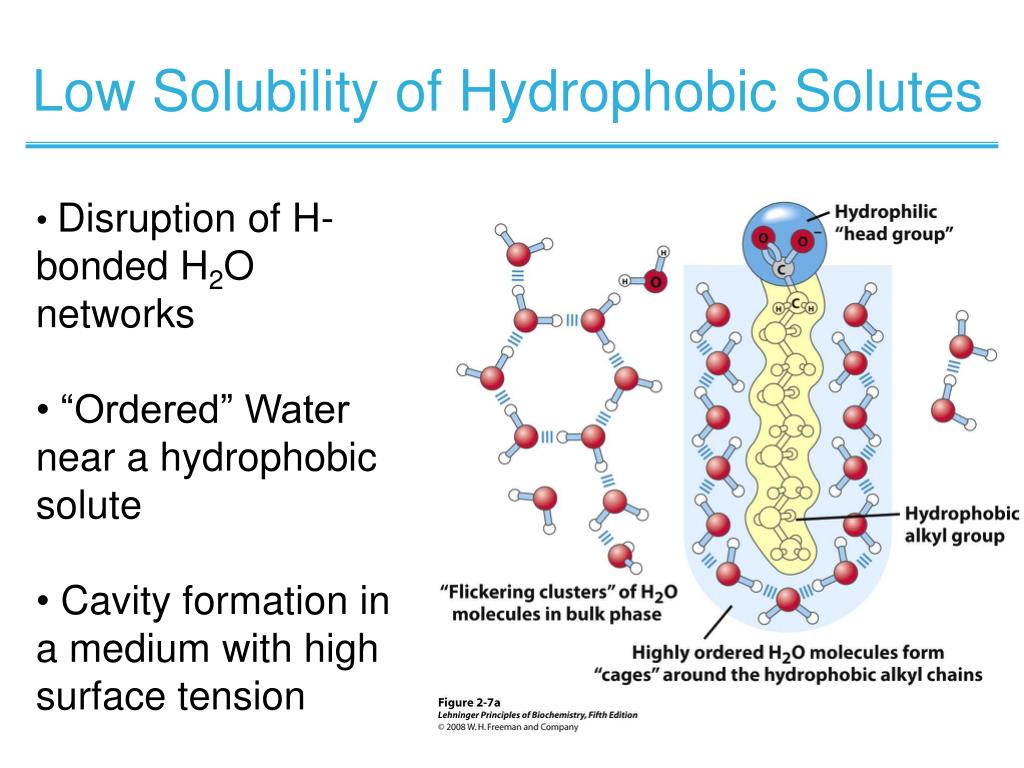 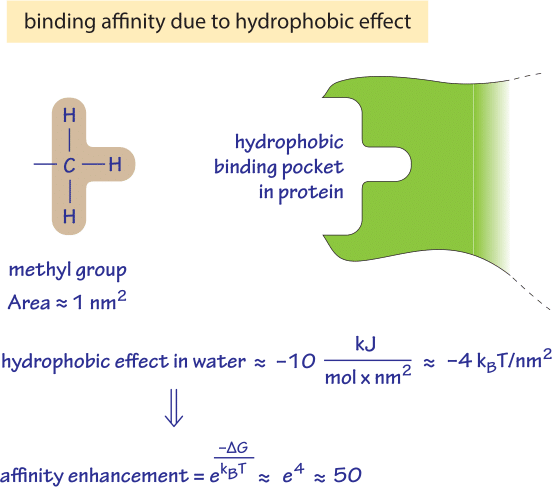
That question can be addressed by referring to an absolute standard, the vapor phase, that neither attracts nor repels solutes, and in which “solute ” molecules exist in isolation most of the time. It is of interest to ask whether nonpolar molecules, such as methane or ethane with no polar groups, tend to leave water and enter less polar solvents (octane is a familiar example) mainly because they are repelled by water or whether they do so because they are attracted to the less polar solvent (or to each other). The physical origins of the hydrophobic effect remain controversial, because the properties of water as a solvent are not yet fully understood. A partial view of the origins of the hydrophobic effect. The tendency of any particular nonpolar molecule or chemical group at equilibrium to favor transfer from water to a nonpolar phase, such as a hydrocarbon solvent, is termed its hydrophobicity. This phenomenon, termed the hydrophobic effect (1, 2), is believed to play a decisive role in maintaining the stability of biological membranes, in the proper folding of protein molecules, and in determining the relative affinities of hormones, antibodies, and substrates for proteins that bind them. In biology, this effect is expressed in the tendency of uncharged (or nonpolar ( molecules to escape from water by entering less polar surroundings, or by adhering to each other (Fig. Cooks and physicists are aware of the tendency of oil to separate from water, a solvent that is very self-cohesive.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
